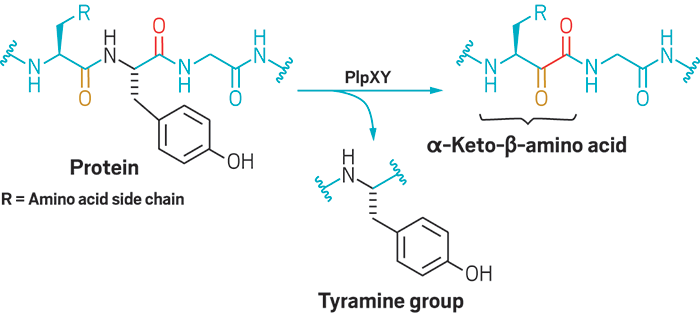
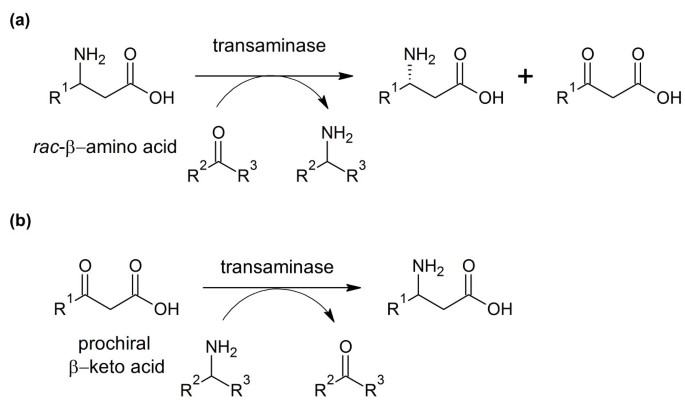
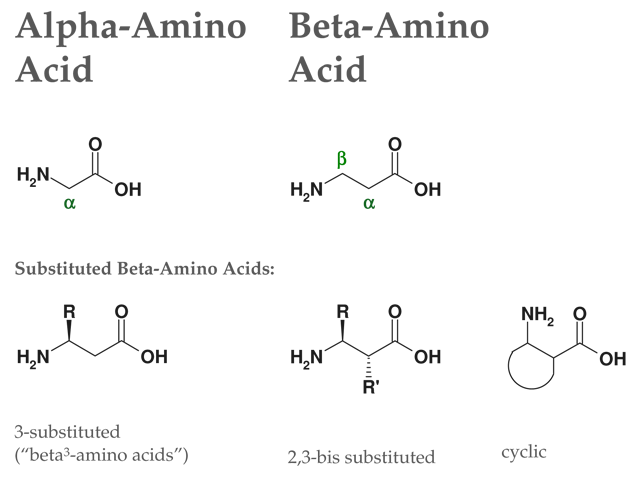
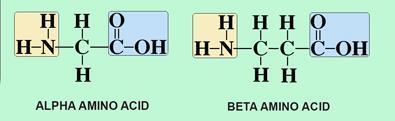
Catalytic, One-Pot Synthesis of β-Amino Acids from α-Amino Acids. Preparation of α,β-Peptide Derivatives | The Journal of Organic Chemistry

Photochemical single-step synthesis of β-amino acid derivatives from alkenes and (hetero)arenes | Nature Chemistry
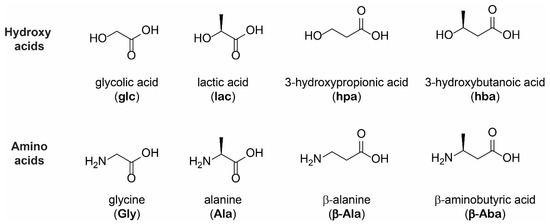
Life | Free Full-Text | Differential Oligomerization of Alpha versus Beta Amino Acids and Hydroxy Acids in Abiotic Proto-Peptide Synthesis Reactions

Photochemical single-step synthesis of β-amino acid derivatives from alkenes and (hetero)arenes | Nature Chemistry
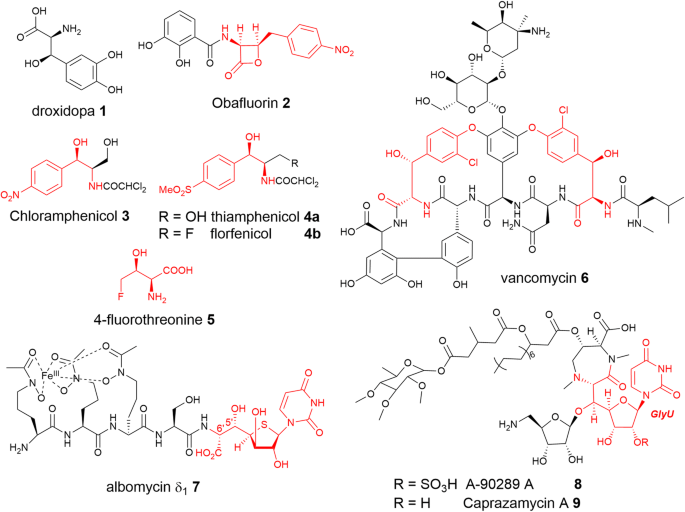
Peculiarities of promiscuous l-threonine transaldolases for enantioselective synthesis of β-hydroxy-α-amino acids | SpringerLink
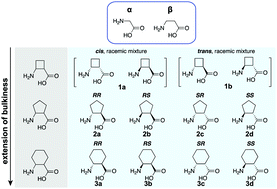
Ribosomal incorporation of cyclic β-amino acids into peptides using in vitro translation - Chemical Communications (RSC Publishing)
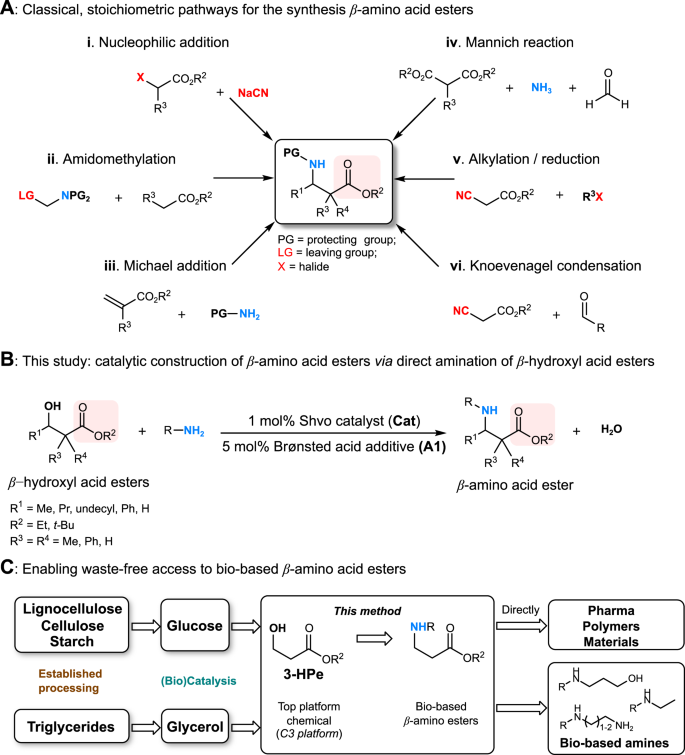
Amination of β-hydroxyl acid esters via cooperative catalysis enables access to bio-based β-amino acid esters | Communications Chemistry

Catalytic, One-Pot Synthesis of β-Amino Acids from α-Amino Acids. Preparation of α,β-Peptide Derivatives | The Journal of Organic Chemistry

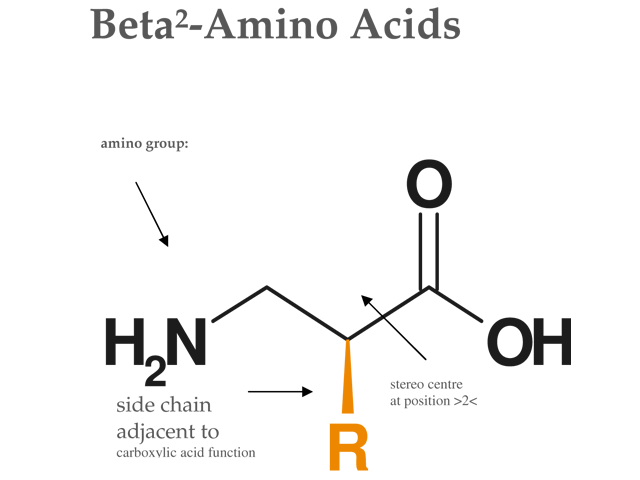
β2‐amino acids—syntheses, occurrence in natural products, and components of β‐peptides1,2 - Lelais - 2004 - Peptide Science - Wiley Online Library

Stereoselective Synthesis of β‐Branched Aromatic α‐Amino Acids by Biocatalytic Dynamic Kinetic Resolution** - Li - 2021 - Angewandte Chemie International Edition - Wiley Online Library